Method development in analytical toxicology
Our department (Department of Forensic Sciences) has a national responsibility to carry out forensic toxicological analyses for the detection of drugs of abuse and medicinal drugs. In forensic toxicology, a continuously expanding range of compounds must be identified and quantified. Since the results of forensic toxicological analysis are used by the judicial system, an unambiguous identification, and accurate and precise quantification, are of paramount importance. Our research group aims to develop analytical methods with high quality and relevant repertoires in common forensic biological matrices. For determination of medicinal and drugs of abuse we mainly use techniques such as UHPLC-MS/MS and UHPLC-QTOF-MS as well as HS-GC-FID for ethanol analysis. We focus on automation and use robots for sample preparation on 96-well plates. Members of the research group also participates in other research projects at Oslo University Hospital (OUH - Research projects). The developed methods are used for both routine analysis of forensic toxicology samples and research projects.
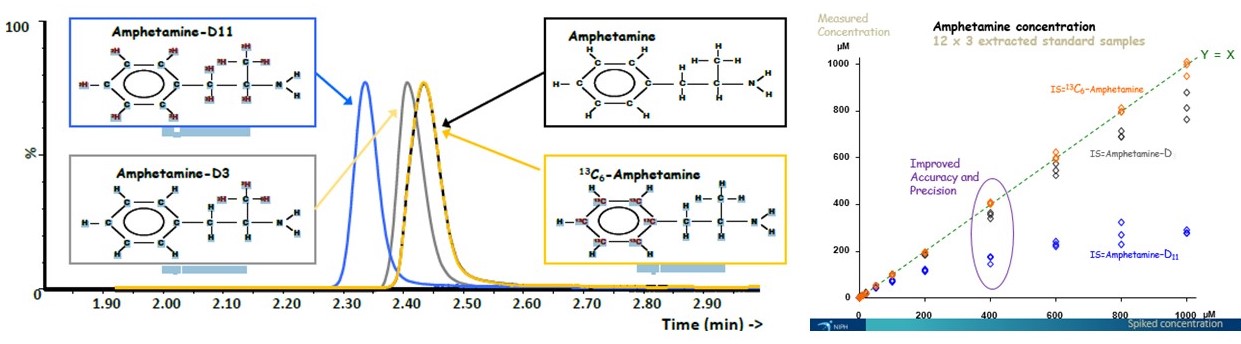
Figure 1. Comparison of deuterium and 13C-labelled internal standards. (T Berg and DH Strand, Oslo University Hospital).
Ongoing projects
UHPLC-MS/MS methods for determination of illegal and medicinal drugs in whole blood, urine or oral fluid by UHPLC-MS/MS
The purpose is to develop and improve screening and confirmation methods using automation of sample preparation on robots and analyses with UHPLC-MS/MS.
UHPLC-QTOF-MS methods for determination of illegal and medicinal drugs
We have been working on developing and improving screening methods using UHPLC-QTOF-MS for many years. The main focus has been on methods for medicinal drugs and new psychoactive substances (NPS) in autopsy samples (whole blood and sometimes muscles), but also methods for detecting NPS in blood samples from police cases as well as in oral fluid from prison service cases.
Development of microextraction methods for forensic applications
Together with the SamplePrep@UiO [Link: SamplePrep@UiO - Department of Pharmacy] group we work with making more sustainable forensic sample preparation methods using liquid-phase microextraction (LPME) and electromembrane extraction (EME). The techniques enables us to reduce the organic solvent consumption to the µl scale and represents greener alternatives to standard sample preparation.
Past projects
Development of a method for analysis of cannabidiol and metabolites for use at the National Centre for Epilepsy
Due to increased interest in the use of cannabidiol in treatment of severe epilepsy, a method for analysis of cannabidiol and metabolites was developed in collaboration with the National Centre for Epilepsy and used to study pharmacokinetic variability and use of therapeutic drug monitoring of cannabidiol in patients with refractory epilepsy.
Development of methods for analysis of medicinal and illicit drugs in biological matrices by UHPLC-MS/MS
We have been working on developing and improving screening and confirmation methods using UHPLC-MS/MS for many years. The main focus has been on methods for medicinal and illicit drugs in forensic whole blood samples (antemortem and postmortem), urine and oral fluid. The necessity for 13C- and 2H-labeled internal standards for the quantitative determination of compounds in biological samples by UHPLC-MS/MS has been shown in several publications (Figure 1).
Development of methods for determination of ethanol and ethanol metabolites
Several methods for determination of ethanol and ethanol metabolites; ethyl glucuronide (EtG), ethyl sulfate (EtS) and phosphatidylethanol (PEth), in forensic samples have been developed. The research group has contributed to other research projects and has developed methods for determination of EtG and EtS in forensic samples (Ethanol and ethanol metabolites and Post-mortem toxicology).
Determination of safety margins for whole blood concentrations of alcohol and nineteen drugs in driving under the influence cases
Measurement uncertainties constitutes of variations on each level in the analytical method; calibration curve, sample withdrawal, sample preparation and chromatographic analysis. In forensic toxicology where there are legislative concentration limits, safety margins have been established in order to ensure that the true concentration is as least as high as the reported concentration, with a 99% safety level. In collaboration with the Norwegian Computing Center (nr.no/en), we developed a model for determination of safety margins for alcohol and nineteen psychoactive drugs.
Comparative evaluation of carboxyhemoglobin quantification in postmortem blood by oximetry and headspace gas chromatography with flame ionization detection and atom absorption spectrophotometry
The HS-GC-FID methods have been regarded as the reference method in forensic toxicology, however, the methods are often complex, time- and sample-consuming, demand dedicated instrumentation and specialized trained analysts. The CO-oximeters require a small sample volume and minimal sample preparation. The CO-oximeters is developed for diagnostic blood gas analyses in hospitals for the 0-20% carboxyhemoglobin (COHb) saturation range. However, in forensic toxicology, the relevant COHb range is from 0-100% saturation. This study compared the carbon monoxide (CO) fractions found in postmortem whole blood samples by using CO-oximetry and HS-GC-FID-AAS. The study found that COHb determination in forensic postmortem whole blood could be based on oximetry analysis alone, with acceptable accuracy, precision, and robustness over the whole range of COHb saturation.
Collaboration
- Stig Pedersen-Bjergaard (professor), Department of Pharmacy, University of Oslo, Norway
- Frederik André Hansen (post doc), Department of Pharmacy, University of Oslo, Norway
- Solfrid Hegstad (Head of section for analytical FOU), Department of Clinical Pharmacology, St. Olav’s Hospital, Norway
- Tonje Gottenberg Skaalvik (Analytical Chemist), Department of Clinical Pharmacology, St. Olav’s Hospital, Norway
- André Fabris (PhD student), School of Pharmaceutical Sciences, University of São Paulo, Brasil
- Maurício Yonamine (Associate Professor), School of Pharmaceutical Sciences, University of São Paulo, Brasil
- Cecile Johannesssen Landmark (professor), National Centre for Epilepsy, Complex Epilepsy Research Group and Therapeutic Drug Monitoring Research Group, Oslo University Hospital
- André Gottås (Head engineer; PhD), National Centre for Epilepsy, Therapeutic Drug Monitoring Research Group, Oslo University Hospital
If you have questions about the projects or suggestions for research collaboration, please contact: Lena Kristoffersen (rmlekr@ous-hf.no) at the Department of Forensic Sciences, Oslo University Hospital, Oslo, Norway.
Publications
Pharmacokinetic variability and use of therapeutic drug monitoring of cannabidiol in patients with refractory epilepsy
Epilepsia (in press)
DOI 10.1111/epi.18284, PubMed 39985501
Dalaker VM, Furuhaugen H, Brekke M, Bjørnaas MA, Krpo M, Øiestad EL, Vallersnes OM (2024)
Drugs in blood and urine samples from victims of suspected exposure to drink spiking: A prospective observational study from Oslo, Norway
PLoS One, 19 (7), e0306191
DOI 10.1371/journal.pone.0306191, PubMed 38985694
Solvent-free parallel artificial liquid membrane extraction for drugs of abuse in plasma samples using LC-MS/MS
Anal Chim Acta, 1301, 342387
DOI 10.1016/j.aca.2024.342387, PubMed 38553114
Skaalvik, TG, Zhou, C, Øiestad, EL, Hegstad S, Trones R and Pedersen‑Bjergaard S (2023)
Conductive vial electromembrane extraction of opioids from oral fluid
Anal Bioanal Chem 415, 5323–5335 (2023).
DOI 10.1007/s00216-023-04807-3, PubMed 37386201/
Skaalvik, TG, Øiestad, EL, Pedersen-Bjergaard, S and Hegstad, S (2023)
Determination of amphetamine enantiomers in urine by conductive vial electromembrane extraction and ultra-high performance supercritical fluid chromatography tandem mass spectrometry
DOI 10.1002/dta.3487, Pubmed 37114617/
Kristoffersen L, Vevelstad M, Hansen I-A, Strømsvåg B and Strand DH (2023)
Comparative Evaluation of Carboxyhemoglobin Quantification in Post-Mortem Whole Blood by CO-Oximetry and Headspace Gas Chromatography with Flame Ionization Detection and Atom Absorption Spectrophotometry
J Anal Toxicol, 47 (3), 311-316 2023.
DOI 10.1093/jat/bkac099, PubMed 36495201/
Sidqey D, Liane VH, Kristoffersen L (2021)
Quantitative Determination of Ethyl Glucuronide and Ethyl Sulfate in Postmortem and Antemortem Whole Blood Using Phospholipid Removal 96-Well Plate and UHPLC-MS-MS
J Anal Toxicol, 45 (4), 378-388
DOI 10.1093/jat/bkaa108 , PubMed 32816025
Schüller M, Tran KTT, Øiestad EL, Pedersen-Bjergaard S (2021)
Membrane-based liquid-phase microextraction of basic pharmaceuticals - A study on the optimal extraction window
J Chromatogr A, 1664, 462769 (in press)
DOI 10.1016/j.chroma.2021.462769, PubMed 34998024
Skaalvik TG, Øiestad EL, Trones R, Pedersen-Bjergaard S, Hegstad S (2021)
Determination of psychoactive drugs in serum using conductive vial electromembrane extraction combined with UHPLC-MS/MS
J Chromatogr B Analyt Technol Biomed Life Sci, 1183, 122926
DOI 10.1016/j.jchromb.2021.122926, PubMed 34624684
Jørgenrud B, Skadberg E, de Carvalho Ponce J, Furuhaugen H, Berg T (2020)
Determination of the alcohol biomarker phosphatidylethanol 16:0/18:1 and 33 compounds from eight different drug classes in whole blood by LC-MS/MS
J Pharmacol Toxicol Methods, 107, 106939
DOI 10.1016/j.vascn.2020.106939, PubMed 33257303
Ask KS, Lid M, Øiestad EL, Pedersen-Bjergaard S, Gjelstad A (2019)
Liquid-phase microextraction in 96-well plates - calibration and accurate quantification of pharmaceuticals in human plasma samples
J Chromatogr A, 1602, 117-123
DOI 10.1016/j.chroma.2019.06.013, PubMed 31229251
Berg T, Eliassen E, Jørgenrud B, Kabashi S, Petukhov A, Bogstrand ST (2018)
Determination of phosphatidylethanol 16:0/18:1 in whole blood by 96-well supported liquid extraction and UHPLC-MS/MS
J Clin Lab Anal, 33 (1), e22631
DOI 10.1002/jcla.22631, PubMed 30047172
Bergh MS, Bogen IL, Wilson SR, Øiestad ÅML (2018)
Addressing the Fentanyl Analogue Epidemic by Multiplex UHPLC-MS/MS Analysis of Whole Blood
Ther Drug Monit, 40 (6), 738-748
DOI 10.1097/FTD.0000000000000564, PubMed 30157097
Kristoffersen L, Langødegård M, Gaare KI, Amundsen I, Terland MN, Strand DH (2018)
Determination of 12 commonly found compounds in DUID cases in whole blood using fully automated supported liquid extraction and UHPLC-MS/MS
J Chromatogr B Analyt Technol Biomed Life Sci, 1093-1094, 8-23
DOI 10.1016/j.jchromb.2018.06.050, PubMed 29980102/
Oiestad AML, Berg T, Eliassen E, Wiklund T, Sand K, Oiestad EL (2018)
Separation of isomers of new psychoactive substances and isotope-labeled amphetamines using UHPSFC-MS/MS and UHPLC-MS/MS
J. Liq. Chromatogr. Relat. Technol., 41 (7), 391-400
DOI 10.1080/10826076.2017.1388818
Takitane J, Leyton V, Andreuccetti G, Gjerde H, Vindenes V, Berg T (2018)
Determination of cocaine, metabolites and a crack cocaine biomarker in whole blood by liquid-liquid extraction and UHPLC-MS/MS
Forensic Sci Int, 289, 165-174
DOI 10.1016/j.forsciint.2018.05.030, PubMed 29885489
Vårdal L, Wong G, Øiestad ÅML, Pedersen-Bjergaard S, Gjelstad A, Øiestad EL (2018)
Rapid determination of designer benzodiazepines, benzodiazepines, and Z-hypnotics in whole blood using parallel artificial liquid membrane extraction and UHPLC-MS/MS
Anal Bioanal Chem, 410 (20), 4967-4978
DOI 10.1007/s00216-018-1147-y, PubMed 29947895
Hegstad S, Kristoffersen L, Liane VH, Spigset O (2017)
EtG and EtS in Autopsy Blood Samples With and Without Putrefaction Using UPLC-MS-MS
J Anal Toxicol, 41 (2), 107-113
DOI 10.1093/jat/bkw123, PubMed 27798076/
Valen A, Leere Øiestad ÅM, Strand DH, Skari R, Berg T (2016)
Determination of 21 drugs in oral fluid using fully automated supported liquid extraction and UHPLC-MS/MS
Drug Test Anal, 9 (5), 808-823
DOI 10.1002/dta.2045, PubMed 27464485
Kristoffersen L, Strand DH, Liane VH, Vindenes V, Tvete IF, Aldrin M (2015)
Determination of safety margins for whole blood concentrations of alcohol and nineteen drugs in driving under the influence cases
Forensic Sci Int, 259, 119-26,
DOI10.1016/j.forsciint.2015.12.009, PubMed 26773222/
Berg T, Karlsen M, Oiestad AM, Johansen JE, Liu H, Strand DH (2014)
Evaluation of ¹³C- and ²H-labeled internal standards for the determination of amphetamines in biological samples, by reversed-phase ultra-high performance liquid chromatography-tandem mass spectrometry
J Chromatogr A, 1344, 83-90
DOI 10.1016/j.chroma.2014.04.020, PubMed 24780257
Eliassen E., Kristoffersen L. (2014)
Quantitative determination of zopiclone and zolpidem in whole blood by liquid-liquid extraction and UHPLC-MS/MS
J. Chromatog B: Anal. Technol. Biomed. Life Sci. 971 (72-80) 2014.
DOI: 10.1016/j.jchromb.2014.09.018, PubMed 25264915/
Amundsen I, Oiestad ÅM, Ekeberg D, Kristoffersen L (2013)
Quantitative determination of fifteen basic pharmaceuticals in ante- and post-mortem whole blood by high pH mobile phase reversed phase ultra high performance liquid chromatography-tandem mass spectrometry
J Chromatogr B Analyt Technol Biomed Life Sci, 927, 112-23
DOI 10.1016/j.jchromb.2012.12.039, PubMed 23380540
Berg T, Jørgenrud B, Strand DH (2013)
Determination of buprenorphine, fentanyl and LSD in whole blood by UPLC-MS-MS
J Anal Toxicol, 37 (3), 159-65
DOI 10.1093/jat/bkt005, PubMed 23423312
Berg T, Strand DH (2011)
¹³C labelled internal standards--a solution to minimize ion suppression effects in liquid chromatography-tandem mass spectrometry analyses of drugs in biological samples?
J Chromatogr A, 1218 (52), 9366-74
DOI 10.1016/j.chroma.2011.10.081, PubMed 22119139
Dahl SR, Olsen KM, Strand DH (2011)
Determination of γ-hydroxybutyrate (GHB), β-hydroxybutyrate (BHB), pregabalin, 1,4-butane-diol (1,4BD) and γ-butyrolactone (GBL) in whole blood and urine samples by UPLC-MSMS
J Chromatogr B Analyt Technol Biomed Life Sci, 885-886, 37-42
DOI 10.1016/j.jchromb.2011.12.009, PubMed 22226469
Karinen R, Øiestad EL, Andresen W, Smith-Kielland A, Christophersen A (2011)
Comparison of the stability of stock solutions of drugs of abuse and other drugs stored in a freezer, refrigerator, and at ambient temperature for up to one year
J Anal Toxicol, 35 (8), 583-90
DOI 10.1093/anatox/35.8.583, PubMed 22004679
Oiestad EL, Johansen U, Oiestad AM, Christophersen AS (2011)
Drug screening of whole blood by ultra-performance liquid chromatography-tandem mass spectrometry
J Anal Toxicol, 35 (5), 280-93
DOI 10.1093/anatox/35.5.280, PubMed 21619723
Sauve EN, Langødegård M, Ekeberg D, Øiestad AM (2011)
Determination of benzodiazepines in ante-mortem and post-mortem whole blood by solid-supported liquid-liquid extraction and UPLC-MS/MS
J Chromatogr B Analyt Technol Biomed Life Sci, 883-884, 177-88
DOI 10.1016/j.jchromb.2011.10.033, PubMed 22119506
Karinen R, Oiestad EL, Andresen W, Wethe G, Smith-Kielland A, Christophersen A (2010)
Comparison of ethanol and other drugs of abuse concentrations in whole blood stored in venoject glass and plastic and venosafe plastic evacuated tubes
J Anal Toxicol, 34 (7), 420-8
DOI 10.1093/jat/34.7.420, PubMed 20822681
Berg T, Lundanes E, Christophersen AS, Strand DH (2008)
Determination of opiates and cocaine in urine by high pH mobile phase reversed phase UPLC-MS/MS
J Chromatogr B Analyt Technol Biomed Life Sci, 877 (4), 421-32
DOI 10.1016/j.jchromb.2008.12.052, PubMed 19144579
Kristoffersen L, Stormyhr LE, Smith-Kielland A (2006)
Headspace gas chromatographic determination of ethanol: the use of factorial design to study effects of blood storage and headspace conditions on ethanol stability and acetaldehyde formation in whole blood and plasma
Forensic Sci Int, 161 (2-3), 151-7
DOI 10.1016/j.forsciint.2006.03.034, PubMed 16843627/
Kristoffersen L, Smith-Kielland A (2005)
An automated alcohol dehydrogenase method for ethanol quantification in urine and whole blood
J Anal Toxicol, 29 (5), 387-9
DOI 10.1093/jat/29.5.387, PubMed 16105266/
