Studies of New Psychoactive Substances
The complexity of the illicit drug market is increasing due to the continuously growing number of new psychoactive substances (NPS). NPS are synthesized with the primary aim of evading existing drug laws and are usually created by modifying the structures of existing psychoactive drugs. They are frequently sold as other common drugs of abuse or mixed in with other drugs, which can lead to accidental overdoses.
To monitor the prevalence of different NPS and enable the acceleration of preventive measures, analysis methods are needed. In forensic casework, the parent compound can be absent or undetectable in biological samples, making metabolite identification essential to confirm the ingested NPS. Knowledge of the pharmacokinetic and pharmacodynamic properties of NPS is important to assess drug potency and the impact of a drug in clinical and forensic toxicology cases.
The primary aim of our research is to develop sensitive and selective analysis methods for the determination of NPS and to expand our knowledge of NPS by studying their metabolism, pharmacodynamics, potency, and mechanism of action.
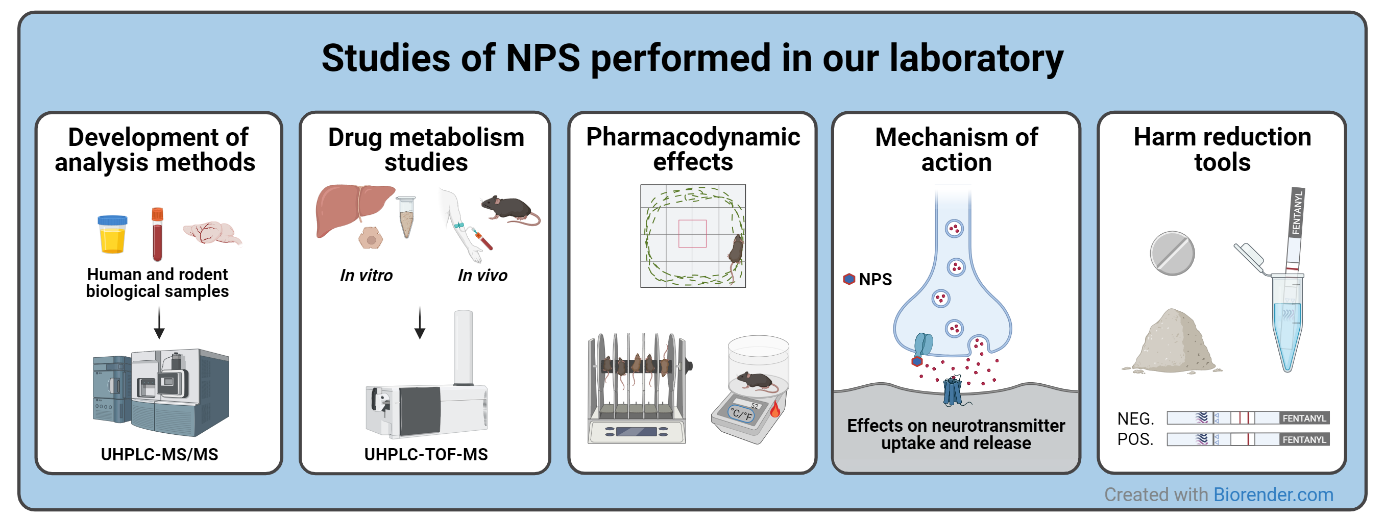
Research Projects
Development of analysis methods
- Develop and validate UHPLC-MS/MS methods for the detection of NPS in different biological matrices
Drug metabolism studies
- Discover the major metabolites of novel fentanyl analogues and synthetic tryptamines using human liver in vitromodels and human post mortem samples combined with UHPLC-TOF-MS
- Study the impact of CYP2D6 genetics for the metabolite pattern of para-methoxymethamphetamine (PMMA) using genotyped human liver microsomes and human post mortem samples
Pharmacologic effects
- Study the pharmacokinetics, pharmacodynamic effects, and potency of selected NPS such as fentanyl analogues, synthetic cathinones, and the methamphetamine analogue methiopropamine
- Study the mechanism of action of new ketamine analogues on the drug market
Harm reduction
- Evaluate the usability of commercially available urine fentanyl test strips to check street drugs for fentanyl or fentanyl analogue contamination
If you have questions about the project or suggestions for research collaboration, please contact: Marianne Skov-Skov Bergh (rmmabe@ous-hf.no) or Inger Lise Bogen (inger.lise.bogen@ous-hf.no) at the Department of Forensic Sciences, Oslo University Hospital, Oslo, Norway.
Publications
Schüller M, McQuade TAP, Bergh MS, Pedersen-Bjergaard S, Øiestad EL (2023)
Determination of tryptamine analogs in whole blood by 96-well electromembrane extraction and UHPLC-MS/MS.
Talanta Open, 7, 100171.
DOI: 10.1016/j.talo.2022.100171
Bergh MS, Bogen IL, Garibay N, Baumann MH (2021)
Pharmacokinetics and pharmacodynamics of cyclopropylfentanyl in male rats
Psychopharmacology (Berl), 238 (12), 3629-3641
DOI 10.1007/s00213-021-05981-x, PubMed 34613431
Bergh MS, Bogen IL, Nerem E, Wohlfarth A, Wilson SR, Oiestad AML (2021)
Discovering the major metabolites of the three novel fentanyl analogues 3-methylcrotonylfentanyl, furanylbenzylfentanyl, and 4-fluorocyclopropylbenzylfentanyl for forensic case work
Forensic Toxicol., 39 (1), 167-178
DOI 10.1007/s11419-020-00560-9
Tuv SS, Bergh MS, Andersen JM, Steinsland S, Vindenes V, Baumann MH, Huestis MA, Bogen IL (2021)
Comparative Neuropharmacology and Pharmacokinetics of Methamphetamine and Its Thiophene Analog Methiopropamine in Rodents
Int J Mol Sci, 22 (21)
DOI 10.3390/ijms222112002, PubMed 34769427
Bergh MS, Øiestad ÅML, Baumann MH, Bogen IL (2020)
Selectivity and sensitivity of urine fentanyl test strips to detect fentanyl analogues in illicit drugs
Int J Drug Policy, 90, 103065
DOI 10.1016/j.drugpo.2020.103065, PubMed 33333419
Gjerde H, Bretteville-Jensen AL, Furuhaugen H, Bache-Andreassen L, Bergh MS, Vindenes V (2020)
Determination of drug residues in used syringe needles
Drug Test Anal, 12 (3), 410-416
DOI 10.1002/dta.2759, PubMed 31899604
Bergh MS, Bogen IL, Garibay N, Baumann MH (2019)
Evidence for nonlinear accumulation of the ultrapotent fentanyl analog, carfentanil, after systemic administration to male rats
Neuropharmacology, 158, 107596
DOI 10.1016/j.neuropharm.2019.04.002, PubMed 30965021
Bergh MS, Bogen IL, Wohlfarth A, Wilson SR, Øiestad ÅML (2019)
Distinguishing Between Cyclopropylfentanyl and Crotonylfentanyl by Methods Commonly Available in the Forensic Laboratory
Ther Drug Monit, 41 (4), 519-527
DOI 10.1097/FTD.0000000000000617, PubMed 30807539
Bergh MS, Bogen IL, Wilson SR, Øiestad ÅML (2018)
Addressing the Fentanyl Analogue Epidemic by Multiplex UHPLC-MS/MS Analysis of Whole Blood
Ther Drug Monit, 40 (6), 738-748
DOI 10.1097/FTD.0000000000000564, PubMed 30157097
Vevelstad M, Øiestad EL, Nerem E, Arnestad M, Bogen IL (2017)
Studies on Para-Methoxymethamphetamine (PMMA) Metabolite Pattern and Influence of CYP2D6 Genetics in Human Liver Microsomes and Authentic Samples from Fatal PMMA Intoxications
Drug Metab Dispos, 45 (12), 1326-1335
DOI 10.1124/dmd.117.077263, PubMed 28978661
Vevelstad M, Øiestad EL, Bremer S, Bogen IL, Zackrisson AL, Arnestad M (2016)
Is toxicity of PMMA (paramethoxymethamphetamine) associated with cytochrome P450 pharmacogenetics?
Forensic Sci Int, 261, 137-47
DOI 10.1016/j.forsciint.2016.02.027, PubMed 26930544
Tuv SS, Bergh MS, Vindenes V, Karinen R (2015)
Methiopropamine in blood samples from drivers suspected of being under the influence of drugs
Traffic Inj Prev, 17 (1), 1-4
DOI 10.1080/15389588.2015.1036157, PubMed 25874580
