A newly identified mitochondrial enzyme protects against obesity.

Anja Solberg (photo), from Arne Klungland's group at the Department of Microbiology, Oslo University Hospital, is first-author on a collaborative work entitled "Deletion of mouse Alkbh7 leads to obesity.", published in Journal of Molecular Cell Biology (journal impact factor 7.667).
Mammals have nine homologues of the Escherichia coli AlkB repair protein: Alkbh1-8, and the fat mass and obesity associated protein FTO. Recently, variants of the FTO gene were identified as obesity risk alleles. Carriers of one copy of a FTO risk allele weighed on average 1 kilo more than those with no copies while carriers of two copies weighed 3 kilo more.
In this collaborative study, Anja Solberg and colleagues, together with collaborators at the Ullevål sykehus (Aronsen and Sjaastad) and NTNU (Rognmo and Wisløff) describe the first functional characterization of mouse ALKBH7. The Alkbh7 protein is located in the mitochondrial matrix (FTO is located in the nucleus) and ALKBH7 deletion dramatically increases body weight and body fat in mice. It is shown that ALKBH7, directly or indirectly, facilitates the utilization of short-chain fatty acids, which might explain the obesity phenotype observed in the ALKBH7 deficient mice.
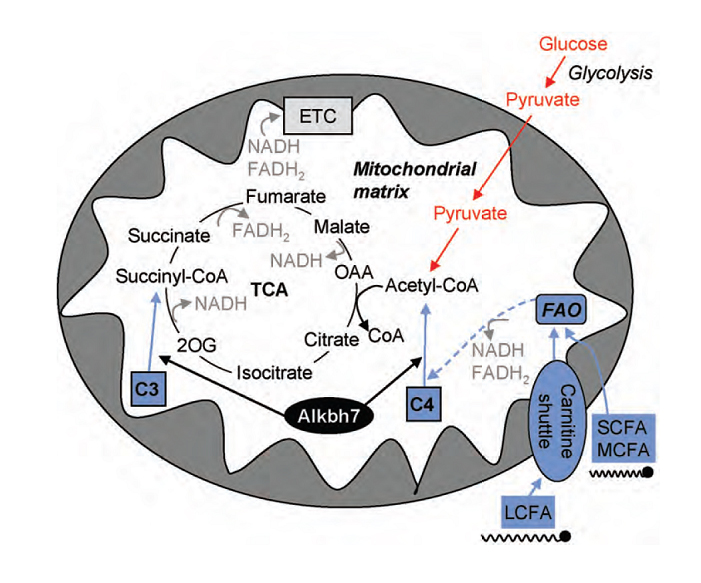 |
| Graphical abstract of paper; Model depicting the proposed role of ALKBH7 in the mitochondria. In this model, ALKBH7 facilitates the entry of the short-chain acylcarnitine species C3 and C4 into the TCA cycle. ALKBH7 might directly or indirectly regulate the conversion of C3 and C4 acylcarnitines to succinyl-CoA and acetyl-CoA, leading to a number of downstream effects in the ALKBH7 deficient mice. Abbreviations; see original paper |
Links:
Deletion of mouse Alkbh7 leads to obesity.
Solberg A, Robertson AB, Aronsen JM, Rognmo O, Sjaastad I, Wisløff U, Klungland A.
J Mol Cell Biol. 2013 Apr 8. [Epub ahead of print]
Arne Klungland's group - Laboratory for Genome repair and regulation
Ivar Sjaastad’s group
Department of Microbiology
