Project group leader Camilla Raiborg Protein dynamics in tumor suppressor pathways

The transformation of a healthy cell into a cancer cell requires multiple events. Knowing the players involved in the processes that regulate cell growth, survival, differentiation, cell division and cell death, can in turn allow us to establish new strategies for diagnosis and treatment of cancers. In our research we focus on protein complexes that shape cellular membranes or alter cellular signalling and in this way contribute to tumour suppression or cancer development. Our aim is to uncover novel cellular mechanisms that can be exploited in cancer medicine.
ESCRTs - endosomal protein complexes required for transport
Nutrients and macromolecules are taken up by cells into small vesicles called endosomes.The endocytic pathway communicates with other membrane-systems in the cell by extensive vesicular transport of membranes and macromolecules between the different compartments. This vesicle traffic serves as a fundamental basis for maintaining cell function and homeostasis, such as nutrient status and signalling output from activated growth factor receptors. Upon activation, signalling receptors are internalized and degraded by the endocytic pathway and this constitutes an important mechanism to attenuate receptor signalling, thus preventing overgrowth. Receptor downregulation is mediated by a huge array of protein complexes commonly known as endosomal protein complexes required for transport (ESCRT). Work from our lab has contributed strongly to the current status of the field. Since the discovery of the ESCRT machinery in 2001/2002, it was not until recently that we’re beginning to understand how components of the ESCRT pathway can serve as prognostic and diagnostic tools in cancer. And still our lab is discovering new ESCRT functions in different cancer relevant cellular processes, lastly in resealing of the nuclear envelope after mitosis. Although we know the constituents of the ESCRT machinery, we completely lack the understanding of how these proteins dynamically work together. In our project group, we have recently obtained groundbreaking new information about this, using cutting edge live-imaging of fluorescently tagged ESCRT-proteins, making ESCRT research one of our main focus areas.
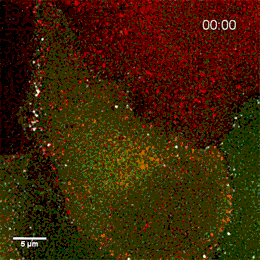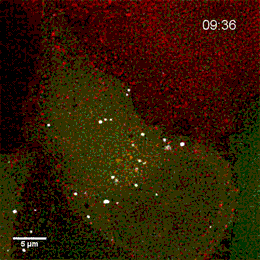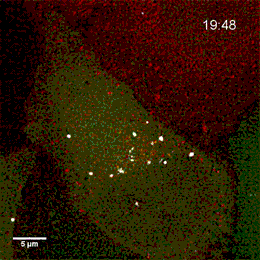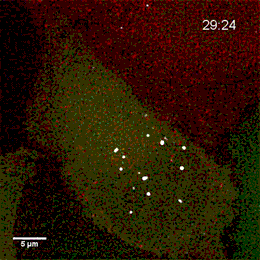
Endosomes containing internalized epidermal growth factor (white)
ER-endosome contact sites
Although the endocytic pathway has been extensively studied for several decades, very little is known about the direct communication between endosomes and the endoplasmic reticulum (ER). In our project group, we have recently identified the basic molecular machinery responsible for the formation of a novel type of ER-endosome contact. We have shown that the spatial proximity of ER and endosomes results in translocation of endosomes to the plasma membrane, where the vesicles fuse with the plasma membrane to form cellular protrusions. This opens up for extensive experimentation on cancer related processes, such as receptor downregulation, autophagy and cell migration. We will continue to explore the composition and function of this new type of contact sites as one of our main focus areas.
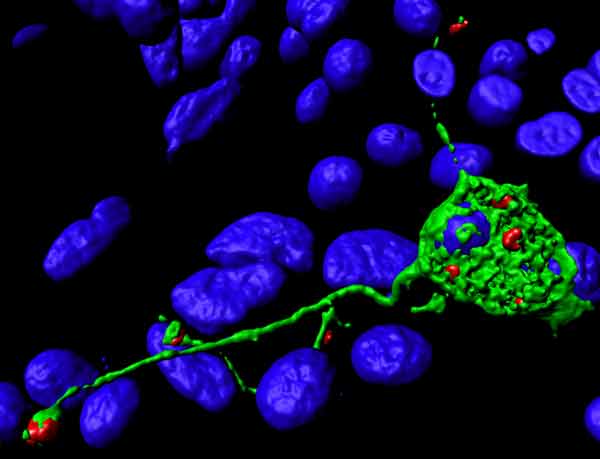
A PC12 cell stimulated with NGF uses late endosomes (red) to grow protrusions. The endoplasmatic reticulum is shown in green.
Link to popular scientific article in Norwegian:
http://forskning.no/celler-kreft/2016/01/filmer-levende-kreftceller
Wnt signalling
The Wnt signalling pathway is central during developmental processes, tissue repair and disease. Inappropriate activity of this pathway results in a variety of human cancers, for example colon cancer. The central molecule of the canonical Wnt signalling pathway is β-catenin, which drives the expression of mitogenic genes, leading to an increased cell proliferation. The protein levels of β-catenin are usually under tight control by the so-called β-catenin destruction complex, which leads to the constitutive degradation of this proto-oncogene in non-proliferative tissue. β-catenin is thereby recruited into a multiprotein destruction complex which includes the structural proteins adenomatous polyposis coli (APC) and axis inhibition protein 1 and 2 (AXIN1/2), and the kinases casein kinase 1α (CK1α) and glycogen synthase kinase 3 (GSK3). The resulting phosphorylation of β-catenin serves as a recognition site for the E3-ligase β-TrCP, which leads to an ubiquitination of β-catenin followed by degradation in the proteasome.
However, the exact interplay of the destruction complex components and the molecular interactions required to process β-catenin remain elusive. For example, the role of APC in the degradation of β-catenin remains poorly understood, but must be significant, as mutations in the APC gene are the most frequent cause of colon cancer. The main goal of our project group is to elucidate the role of the β-catenin destruction complex in cancer development by molecularly dissecting its composition, structure, function and regulation.

Cover of November edition of Molecular Cancer Research 2015 by Thorvaldsen, Pedersen, Wenzel et al.
3D structured illumination micrograph of SW480 colon carcinoma cells showing Tankyrase-inhibitor induced degradasomes.
| Our research is supported by : |  |
|
|
Coworkers:
Key publications ESCRTs:
Wenzel EM, Schultz SW, Schink KO, Pedersen NM, Nähse V, Carlson A, Brech A, Stenmark H, Raiborg C (2018) Concerted ESCRT and clathrin recruitment waves define the timing and morphology of intraluminal vesicle formation. Nat Commun, 9 (1), 2932 DOI 10.1038/s41467-018-05345-8, PubMed 30050131
Vietri M, Schink KO, Campsteijn C, Wegner CS, Schultz S, Christ L, Thoresen SB, Brech A, Raiborg C and Stenmark H. (2015) Spastin and ESCRT-III coordinate mitotic spindle disassembly and nuclear envelope sealing. Nature. Jun 11;522(7555):231-5
- Comment in Sundquist WI and Ullman KS. (2015) An ESCRT to seal the envelope. Science. Jun 19;348(6241)
- Comment in Burk B. (2015) Nuclear dilemma resolved. Nature. Jun 11;522(7555)
- This article was recommended by the Faculty of 1000: http://f1000.com/prime/725533850
Raiborg C and Stenmark, H. (2009) The ESCRT machinery in endosomal sorting of ubiquitylated membrane proteins. Nature. Mar 26;458(7237):445-52.
Raiborg C, Malerød L, Pedersen NM, Stenmark H. (2008) Differential functions of Hrs and ESCRT proteins in endocytic membrane trafficking. Exp Cell Res. Feb 15;314(4):801-13.
- Cover on Exp Cell Res2009 on line edition.
Raiborg C, Wesche J, Malerod L, Stenmark H. (2006) Flat clathrin coats on endosomes mediate degradative protein sorting by scaffolding Hrs in dynamic microdomains. J Cell Sci. Jun 15;119:2414-24
Raiborg C, Rusten TE, Stenmark H. (2003) Protein sorting into multivesicular endosomes. Curr. Opin. Cell Biol. Aug;15(4):446-55.
Raiborg C, Bache KG, Gillooly DJ, Madshus IH, Stang E, Stenmark H. (2002) Hrs sorts ubiquitinated proteins into clathrin-coated microdomains of early endosomes. Nat Cell Biol. 4:394-8.
- Comment in Clague MJ. (2002) Membrane transport: a coat for ubiquitin. Curr. Biol. 12: 529-31.
- Comment in Finly D. (2002) Ubiquitin chained and crosslinked. Nat. Cell. Biol. 4: 121-3.
Raiborg C, Bache KG, Mehlum A, Stang E, Stenmark H. (2001) Hrs recruits clathrin to early endosomes. EMBO J. 20(17):5008-21.
Raiborg C,Bremnes B, Mehlum A, Gillooly DJ, D'Arrigo A, Stang E, Stenmark H. (2001) FYVE and coiled-coil domains determine the specific localisation of Hrs to early endosomes. J Cell Sci.. 114(12):2255-63.
Key publications ER-endosome contact sites:
Hong Z, Pedersen NM, Wang L, Torgersen ML, Stenmark H, Raiborg C (2017) PtdIns3P controls mTORC1 signaling through lysosomal positioning. J Cell Biol, 216 (12), 4217-4233 DOI 10.1083/jcb.201611073, PubMed 29030394
Raiborg C, Wenzel EM and Stenmark H. (2015) ER-endosome contact sites: molecular compositions and functions. EMBO J. Jul 14;34(14):1848-58
Raiborg C*, Wenzel EM, Pedersen NM, Olsvik H, Schink KO, Schultz SW, Vietri M, Nisi V, Bucci C, Brech A, Johansen T and Stenmark H*. (2015) Repeated ER-endosome contacts promote endosome translocation and neurite outgrowth. Nature. Apr 9;520(7546):234-8 * Corresponding authors
- Comment in Krauss and Haucke. (2015) A grab to move on: ER-endosome contacts in membrane protrusion formation and neurite outgrowth. EMBO J. Jun 3;34(11):1442-4
- Comment in Wijdeven RH et al.,(2015) ER contact sites direct late endosome transport. Bioessays 37 in press.
- This article was recommended by the Faculty of 1000: http://f1000.com/prime/725428494
Key publications Wnt signalling:
Thorvaldsen TE, Pedersen NM, Wenzel EM, Schultz SW, Brech A, Liestøl K, Waaler J, Krauss S, Stenmark H. Structure, Dynamics, and Functionality of Tankyrase Inhibitor-Induced Degradasomes. Mol Cancer Research 2015, 13(11):1487-501
- Cover on November edition of Molecular Cancer Research
Schneikert J, Ruppert JG, Behrens J, Wenzel EM, Different Roles of Axin Interacting with the SAMP and Second Twenty Amino Acid Repeats of Adenomatous Polyposis Coli. PLoS One. 2014, 9(4):e94413
- This article was recommended by the Faculty of 1000: http://f1000.com/prime/718346651
Kohler EM, Chandra SH, Behrens J, Schneikert J, Beta-catenin degradation mediated by the CID domain of APC provides a model for the selection of APC mutations in colorectal, desmoid and duodenal tumours. Hum Mol Genet 2010, 18(2): 213-226
Kohler EM, Brauburger K, Behrens J, Schneikert J, Contribution of the 15 amino acid repeats of truncated APC to beta-catenin degradation and selection of APC mutations in colorectal tumours from FAP patients. Oncogene 2009, 29(11): 1663-1671
Kohler EM, Derungs A, Daum G, Behrens J, Schneikert J, Functional definition of the mutation cluster region of adenomatous polyposis coli in colorectal tumours. Hum Mol Genet 2008, 17(13): 1978-1987



