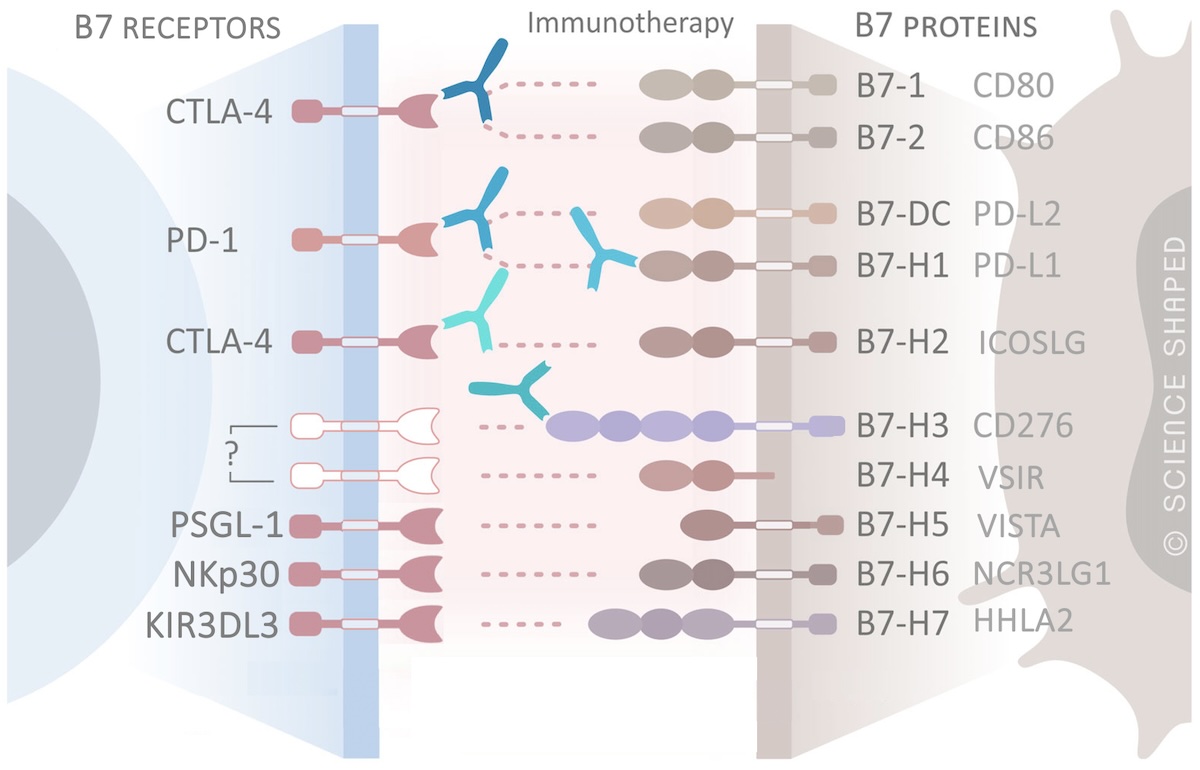
B7 immune checkpoint proteins as novel therapeutic targets in cancer

Immunotherapies targeting immune checkpoint proteins are therapeutic approaches that mobilize the immune system to kill the cancer cells. Current therapies use monoclonal antibodies that block receptor-ligand interactions and prevent inhibitory actions on the anti-tumor immune response. Even though a group of patients respond to therapy and obtain long-lasting responses, many patients do not respond or develop therapy resistance, emphasizing the need for novel and more effective treatments. To develop new alternative and improved immunotherapies, it is critical to understand the molecular mechanisms underlying the functional activity of the immune checkpoint proteins. One of our research interests is to validate the alternative B7 immune checkpoint proteins as novel biomarkers and therapeutic targets in cancer, by unraveling their functions and their relation to tumorigenesis.
We have contributed knowledge on the Impact of B7-H3 expression on metastasis, immune exhaustion and JAK/STAT and PI3K/AKT pathways in clear cell renal cell carcinoma, miRNA regulating B7-H3, and its clinical implications in breast cancer. We have also deciphered the role of B7-H3 in glycolysis and sensitivity to chemotherapy and targeted drugs in breast cancer and melanoma, as well as discovered molecular mechanisms underlying the chemo-sensitive role of B7-H3. We have further described correlation of B7-H3 expression with poor outcome, Major Vault Protein, and androgen receptor in prostate cancer, and provided important knowledge on the functional role of glycosylated B7-H5/VISTA in metastatic renal cancer, and on B7-H4 in sensitivity of renal cancer cells to targeted therapies.
You can read more about our recent findings in this blog post and Editorial, and in our reviews on B7-H3 in human cancer beyond immune regulation, on the hopes of targeting B7-H3 in neuroblastoma, and B7-H3: a robust target for immunotherapy in prostate cancer.