Intratumor heterogeneity
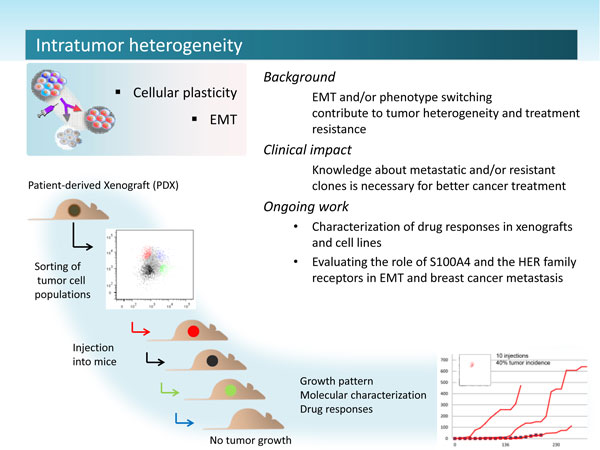 |
EMT
The process of epithelial to mesenchymal transition, where epithelial cells loose polarity and gain motility is important in developmental processes in the embryo, in breast development, lung development, and during wound healing. The metastasis promoting protein S100A4 has previously been shown to be upregulated in epithelial cells after EMT and has been suggested to be a mesenchymal marker. It is suggested that carcinoma cells harboring the ability to undergo EMT and the reverse process MET have the highest ability to escape therapy and form secondary tumors. EMT is thought of as a trans-differentiation process, and trans-differentiating cells within a tumor mass contribute to the complex intratumor heterogeneity of solid cancers.
Intratumor heterogeneity
To understand how some cancer cells are able to escape and cause recurrence, researchers have compared primary tumors to small ecosystems where the extracellular components determine the physical environment, and all cell populations, both normal and neoplastic, represent the diversity of the species within the system. Large intratumor cellular diversity ensures that at least one tumor cell subpopulation is able to tolerate the altered conditions, during treatment, or relocation to a metastatic site. Our challenge is to understand why heterogeneity is sustained in the developing tumor, and how to best eradicate dynamically changing cancer cell populations before they develop strategies to withstand attacks from various treatment. Intratumor heterogeneity caused by genetic, phenotypic or functional differences between cancer cell subpopulations is a considerable clinical challenge. Understanding subpopulation dynamics is therefore central for both optimization of existing therapy and for development of new treatment.